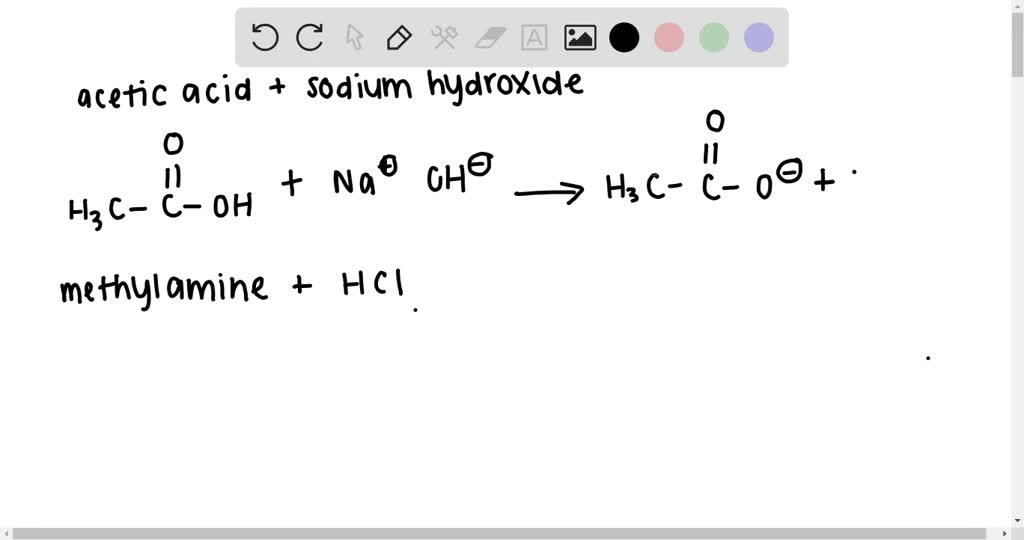
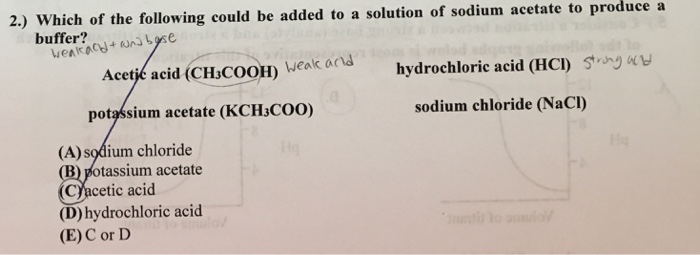ACIDS, BASES, AND SALTS. THE IMPORTANCE OF ACIDS, BASES, AND SALTS Almost all inorganic compounds and many organic compounds can be classified as acids, - ppt download

Sequential Acetic Acid–Sodium Chloride Treatment to Control Salt Stoichiometry of a Hydrochloride Salt | Organic Process Research & Development

Sequential Acetic Acid–Sodium Chloride Treatment to Control Salt Stoichiometry of a Hydrochloride Salt | Organic Process Research & Development

Sodium Chloride - Acetic Acid Standard, 2.00% (w/w) Sodium Chloride/1.00% (w/w) Acetic Acid, Ricca Chemical, Quantity: 4 L | Fisher Scientific

Show the dissociation of the following compounds on dissolving in water, with the help of chemical equation and write whether the proportion of dissociation is small or large.Hydrochloric acid, Sodium chloride, Potassium

Correlation between concentration and electrical conductivity (EC) of... | Download Scientific Diagram

SOLVED: 3) Select the net ionic equation for the reaction that occurs when sodium chlorlde and acetic acid are mlxed No reaction DCCuT (aa) Cl (aq) H' (aq) CzlL,O. (an) (aw) C,H,O; (
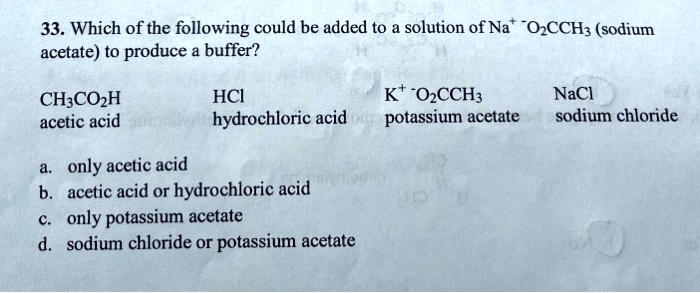
SOLVED: 33. Which of the following could be added to solution of Na OzCCH; (sodium acetate) to produce buffer? CH;COzH acetic acid HCI hydrochloric acid Kt -OCCH3 potassium acetate NaCl sodium chloride

SOLVED: Consider the following species: hydrochloric acid, acetic acid, sodium chloride and glucose. Select all answers that are correct A.)Acetic acid and glucose are weak electrolytes B.)Three of the four species will

The equivalent conductances of sodium chloride, hydrochloric acid and sodium acetate at infinite dilution are 126.45, 426.16 and 91.0 ohm^(-1) cm^(2) eq^(-1), respectively at 25^(@)C. Calculate the equivalent conductance of acetic acid