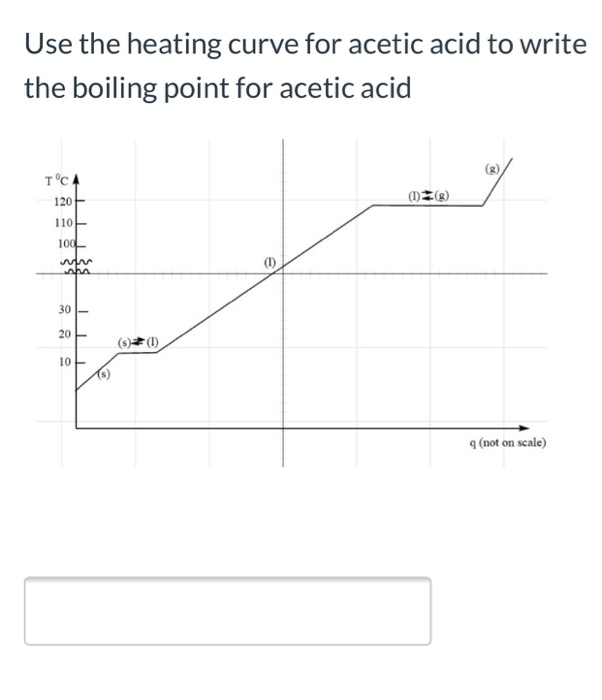
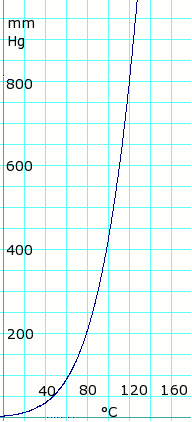
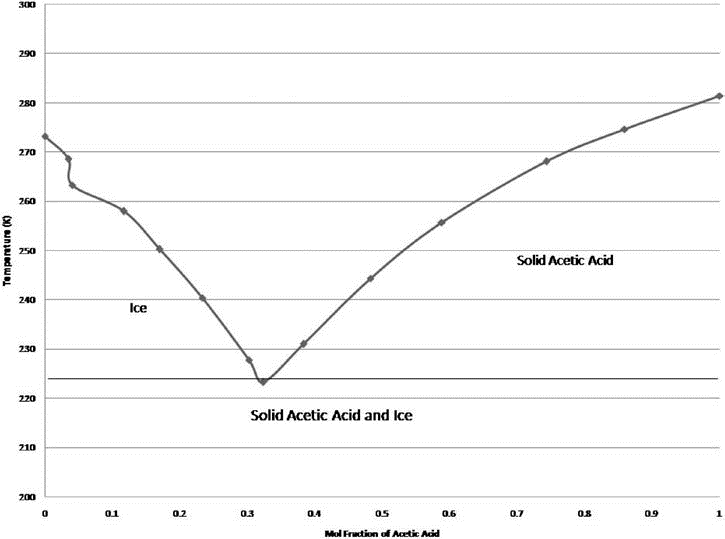
⚗️The melting point of acetic acid is 17°C, and the boiling point is 118°C. Draw a graph showing the - Brainly.com
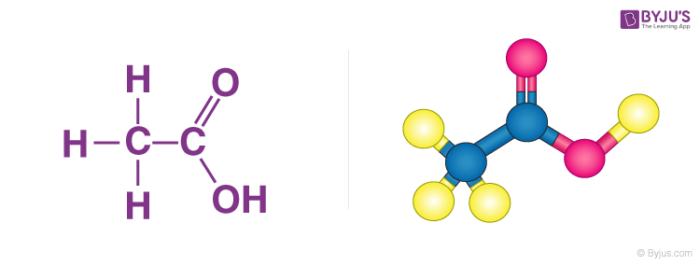
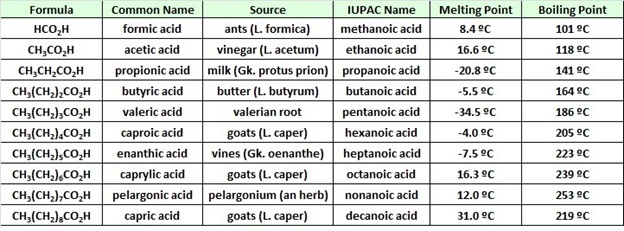
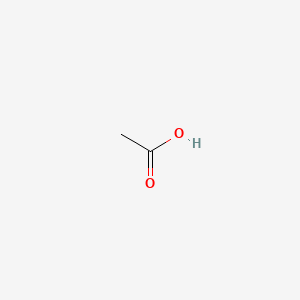
Acetic Acid (CH3COOH)- Structure, Properties, Preparation, Physical, Chemical properties, Uses and FAQs of Acetic acid

2-aminooxy-acetic acid - 2921-14-4, C2H5NO3, density, melting point, boiling point, structural formula, synthesis
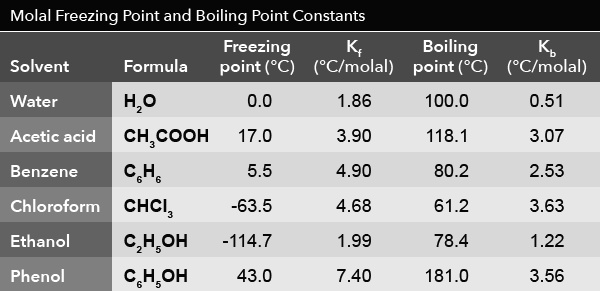
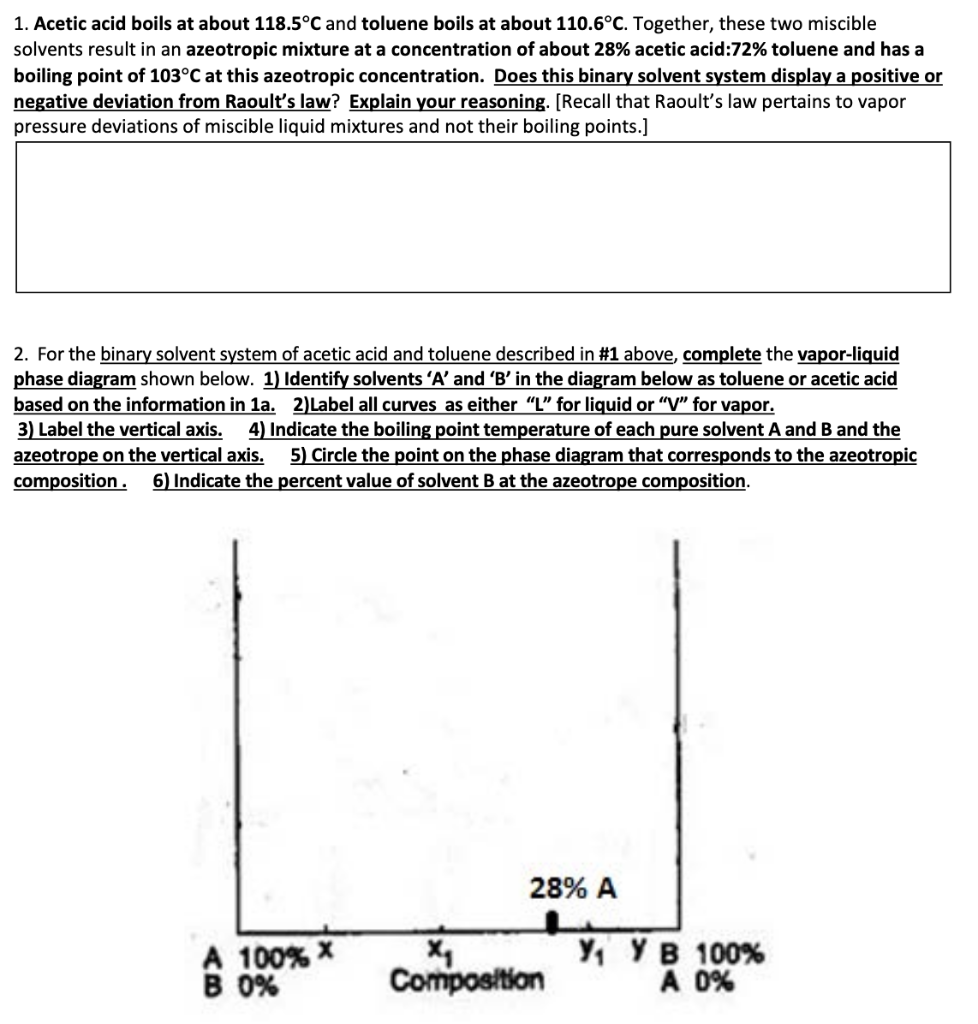
SOLVED: Molal Freezing Point and Boiling Point Constants Freezing Boiling Kb Solvent Formula point (PC) ('Clmolal) point (PC) (Clmolal) Water Hzo 0.0 Acetic acid CH;COOH 17.0 Benzene Cehs 5.5 Chloroform CHCI -63.5

Arrange the following compounds in increasing order of their boiling points:acetic acid, methyl formatae, acetamide, propan - 1 - ol