Copper based catalysts for the selective ammonia oxidation into nitrogen and water vapour—Recent trends and open challenges - ScienceDirect

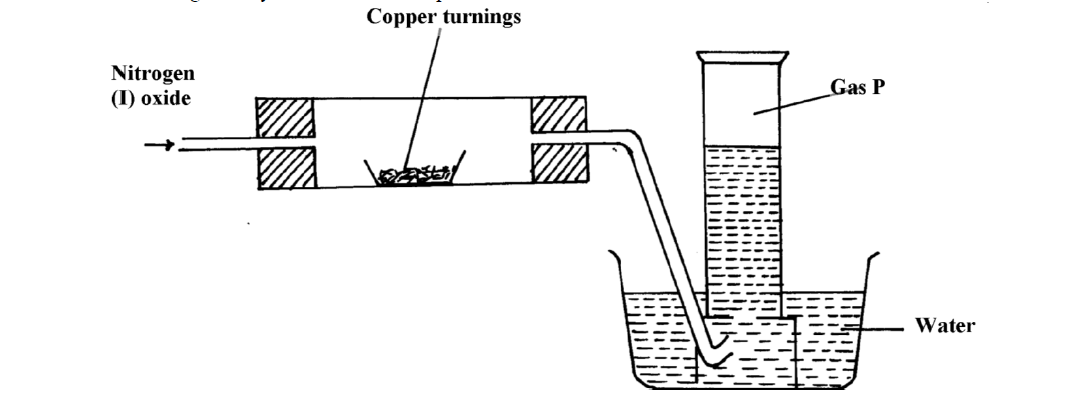
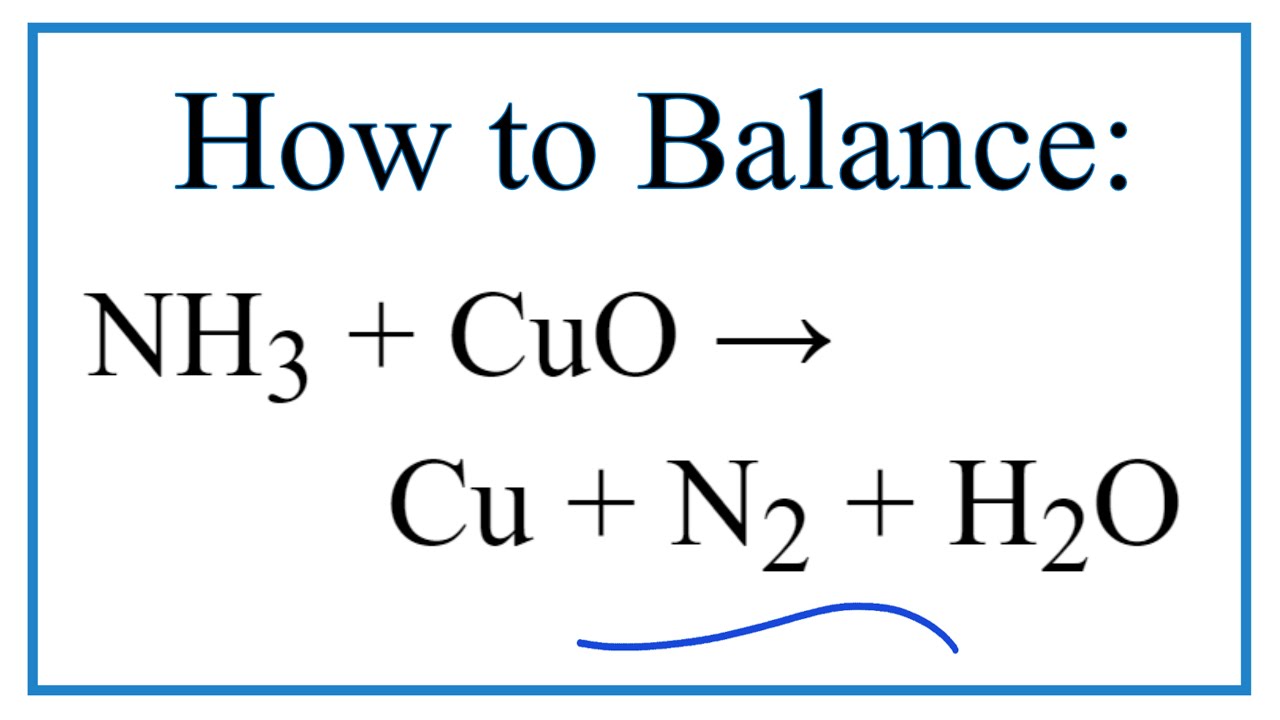
When ammonia is passed over heated copper oxide, the metallic coper is obtained. The reaction shows - YouTube

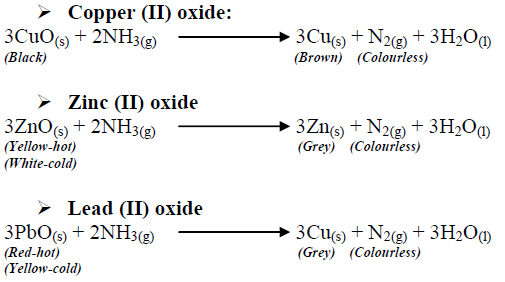
Give balanced equation for each of the following:Reduction of hot Copper (II) oxide to copper using ammonia gas:

Electrocatalytic, Homogeneous Ammonia Oxidation in Water to Nitrate and Nitrite with a Copper Complex | Journal of the American Chemical Society

When ammonia is passed over heated copper oxide, the metallic copper is obtained, the reaction shows - YouTube

When ammonia is passed over heated copper oxide, the metallic copper is obtained, the reaction shows - YouTube
Dry ammonia gas was passed over heated lead (II) oxide and the products passed over anhydrous copper - Tutorke

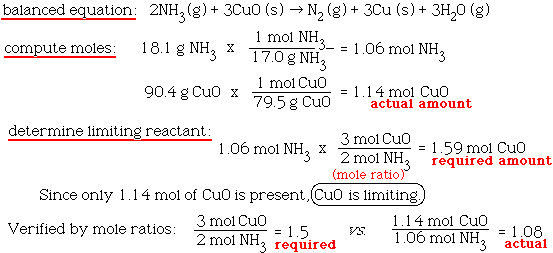
SOLVED: How many moles of nitrogen gas would be produced if 3.40 moles of copper(II) oxide were reacted with excess ammonia in the following chemical reaction? 2 NHz(g) + 3 CuO (s) -

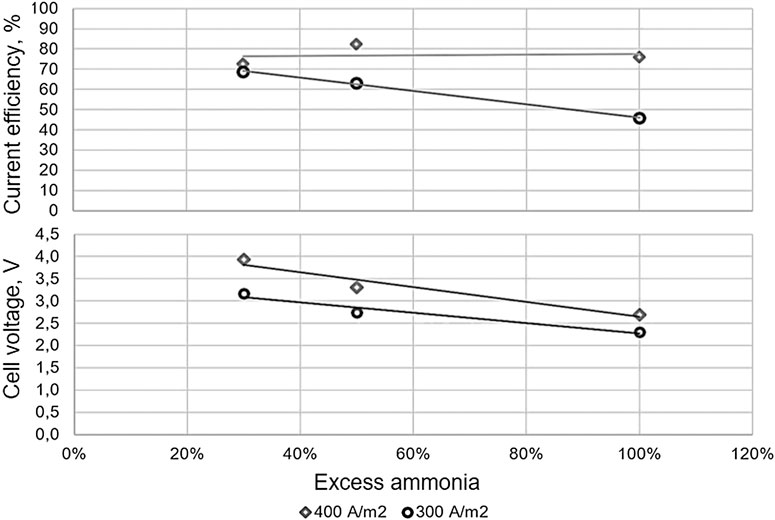
Coatings | Free Full-Text | Electro-Oxidation of Ammonia over Copper Oxide Impregnated γ-Al2O3 Nanocatalysts

Activating copper oxide for stable electrocatalytic ammonia oxidation reaction via in-situ introducing oxygen vacancies | SpringerLink
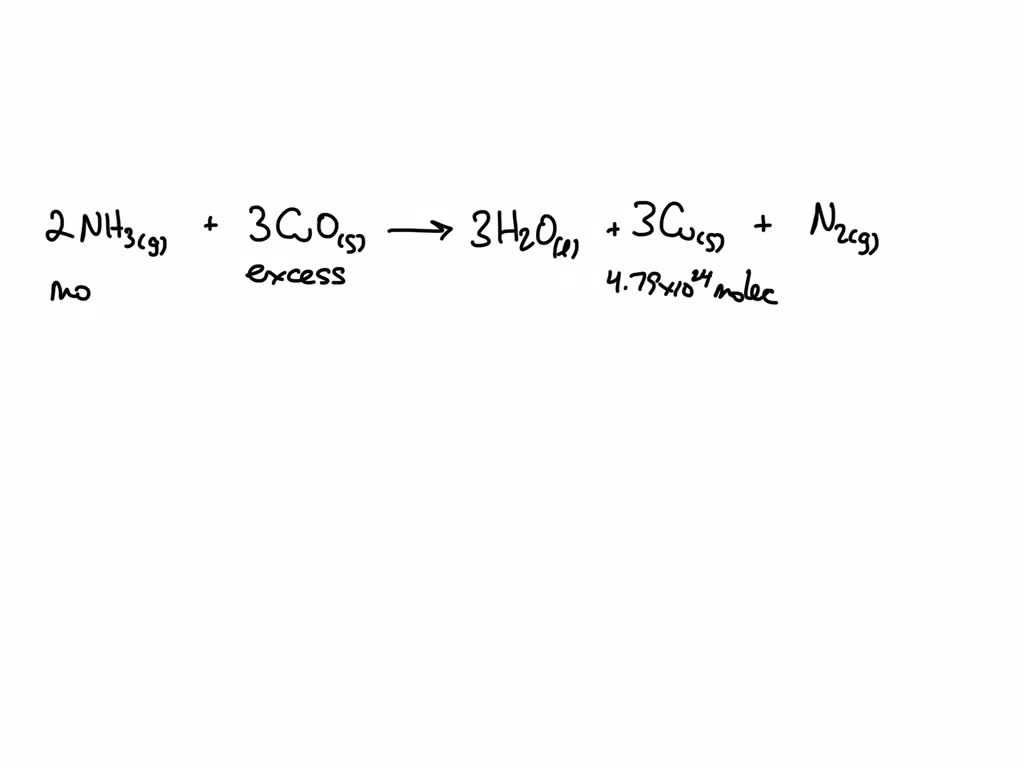
SOLVED: Ammonia gas and solid copper(II) oxide react to form liquid water, solid copper, and nitrogen gas. If copper(II) oxide is present in excess, how many moles of ammonia are needed to

Coatings | Free Full-Text | Electro-Oxidation of Ammonia over Copper Oxide Impregnated γ-Al2O3 Nanocatalysts
![PDF] Dissolution kinetics of low grade complex copper ore in ammonia- ammonium chloride solution | Semantic Scholar PDF] Dissolution kinetics of low grade complex copper ore in ammonia- ammonium chloride solution | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/74104461ce1759e6c90926cb2fd02cdd1b4c9a35/2-Table1-1.png)
PDF] Dissolution kinetics of low grade complex copper ore in ammonia- ammonium chloride solution | Semantic Scholar

SOLVED: 1) Ammonia and copper (II) oxide react according to the following equation: 2NH3 + 3 CuO → N2 + 3 Cu + 3 H2O if 57.0 g of ammonia are combined