A company manufactures car batteries of a particular type. The lives (in years) of 40 such batteries were recorded as follows: 2.6 3.0 3.7 3.2 2.2 4.1 3.5 4.5 | 3.5 2.3

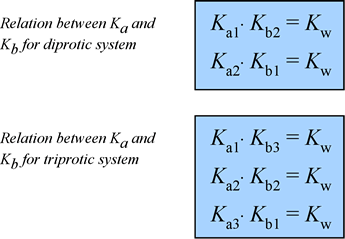
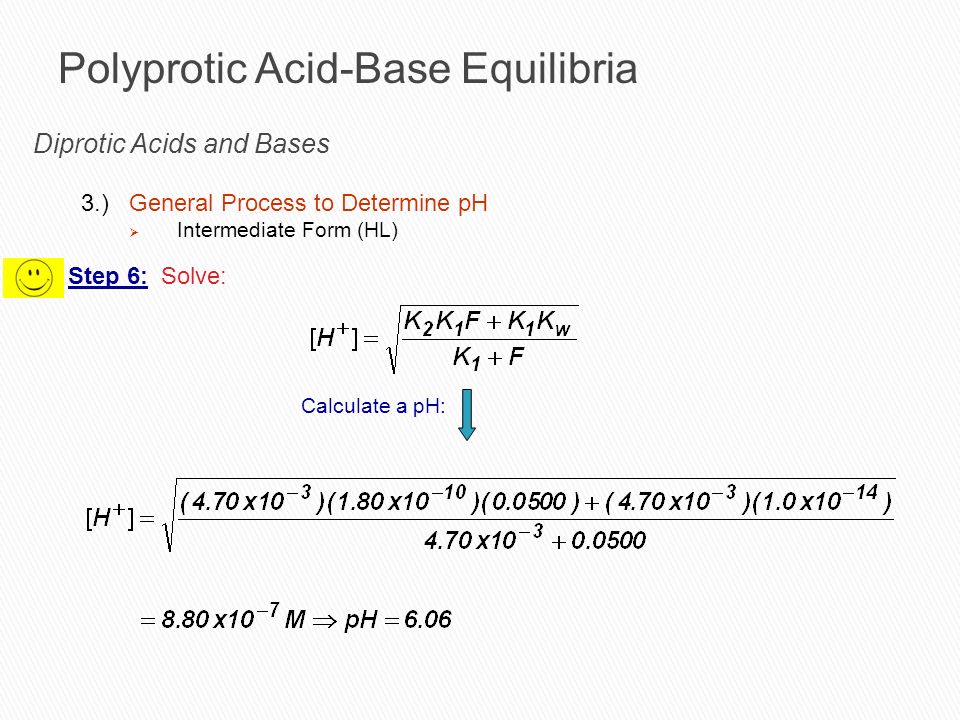
pH Calculation of mixture of weak acids, bases, diprotic and triprotic weak acids | pH calculation of amphiprotic salts
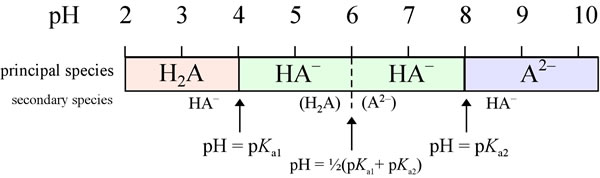
First, notice that the pH where two species concentrations are the same is around the pKa for that equilibrium. In fact, for polyprotic acids with. - ppt download

equilibrium - Why do the first and second ionizations of a diprotic acid produce the same hydronium ion concentrations in solution? - Chemistry Stack Exchange