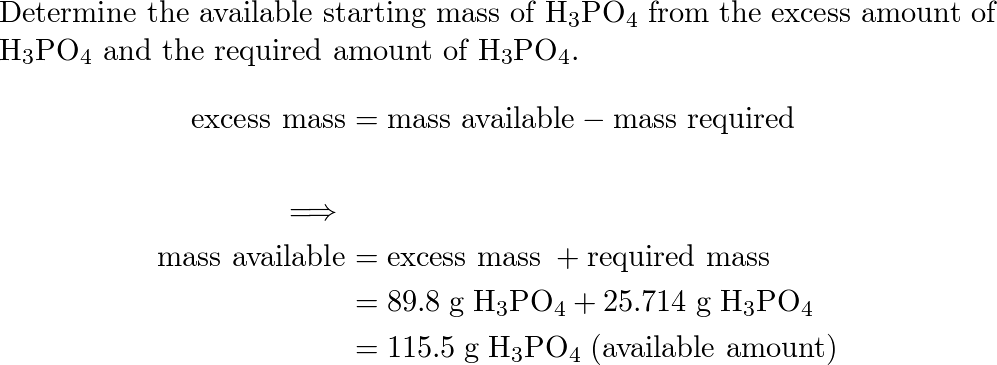H3PO4+KOH=K3PO4+H2O Balanced Equation||Phosphoric acid+Potassium hydroxide=Potassium phosphate+Water - YouTube

Ch19.1 – Acids and Bases Acids - corrosive, taste sour, put electrolytes in soln, react with metals Ex1) Single displacement reaction: H 2 SO 4(aq) + Zn. - ppt download
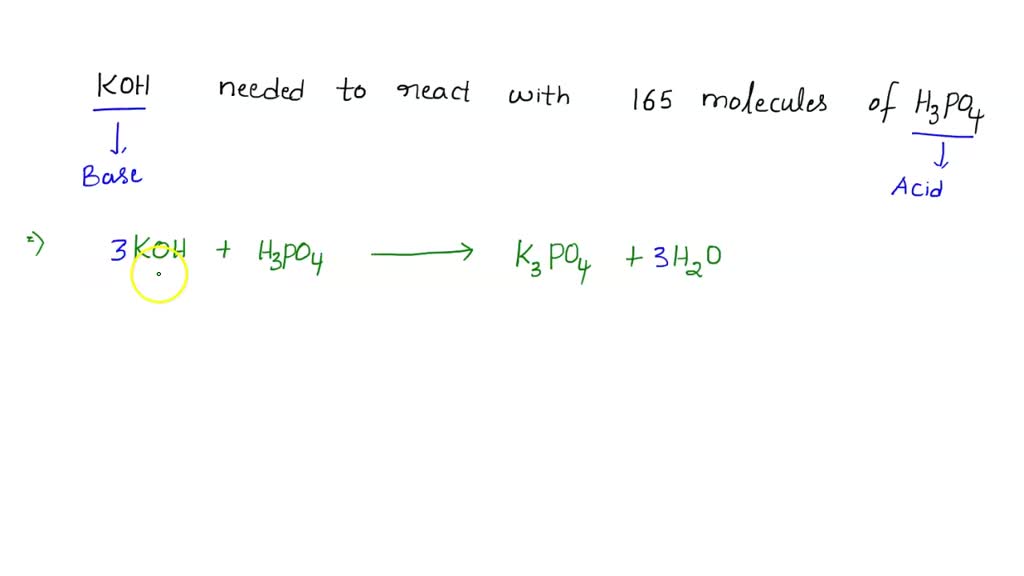
SOLVED: 'How many units of potassium hydroxide are needed to react with 165 molecules of phosphoric acid? BKOHq) HzPO4aq) KzPO4aq) 3HzQ()'

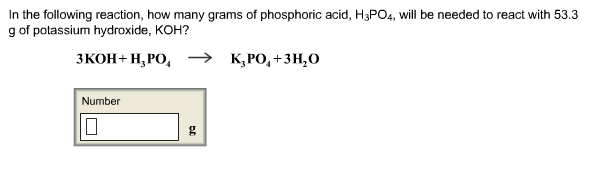
SOLVED: 97. This reaction shows the neutralization of phosphoric acid (H;POA) with potassium hydroxide (KOH): H3PO4(aq)+3 KOH (aq)K3PO4(aq+3 H2O (4) HzPOa (aq) + 3KOH (aq) = K:POa (aq) + 3Hz0 (U) If

SOLVED: Question 32 (3 points) Which of the following represents the molecular equation for the complete neutralization reaction of phosphoric acid (HzPOA) with aqueous potassium hydroxide (KOH)? HyPOa (aq) KOH(aq) Hzo() KHzPOA(aq)
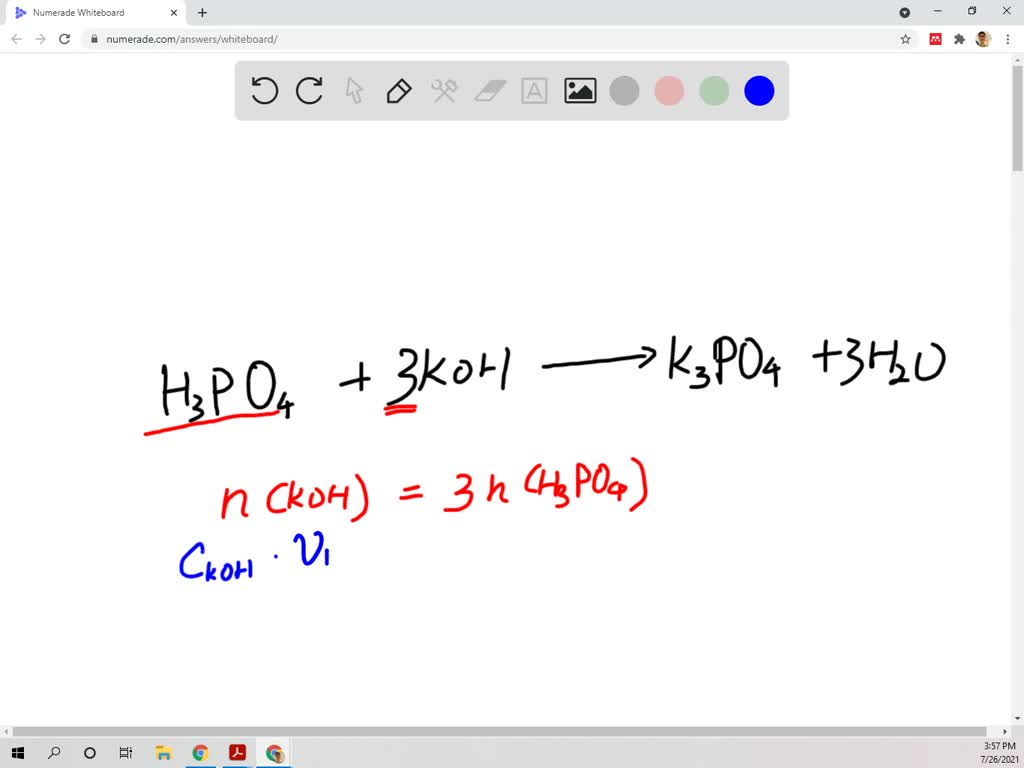
SOLVED: A 0.0700 L sample of phosphoric acid (H3PO4) solution with an unknown concentration reacts with 0.200 L of 0.300 M potassium hydroxide ( KOH) solution. What is the concentration of the phosphoric
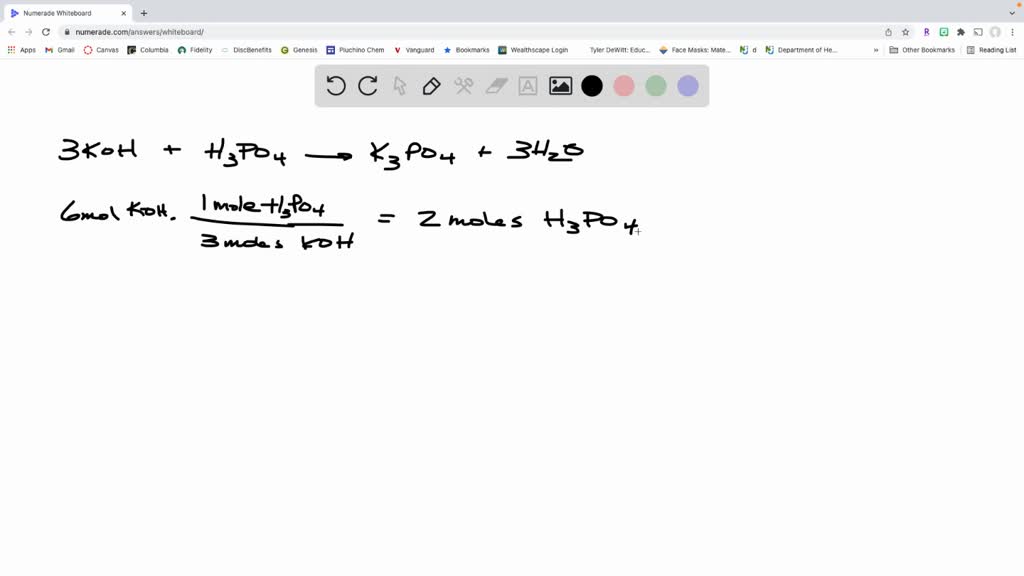
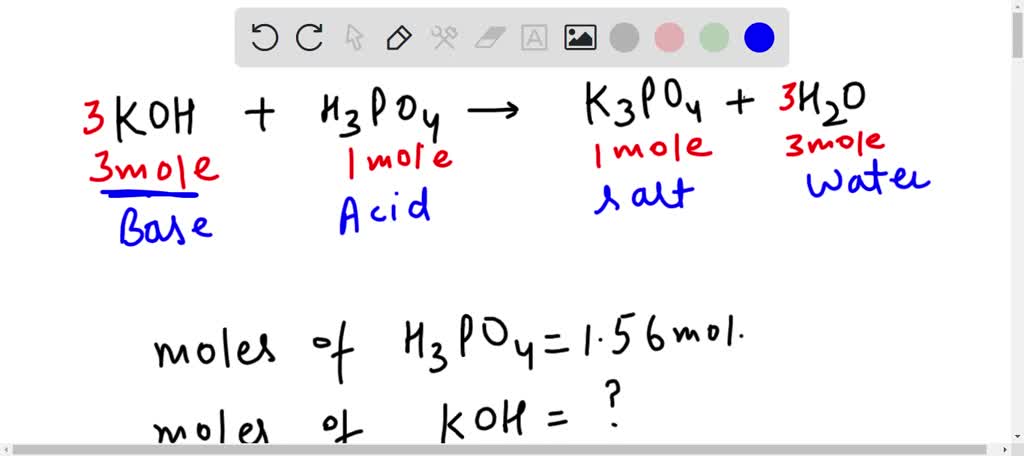
SOLVED: When potassium hydroxide reacts with phosphoric acid, potassium phosphate and water are produced. The balanced equation for this reaction is: 3KOH(aq) + H3PO4 (aq) -> K3PO4(aq) + 3H2O(l) If 6 moles

SOLVED: 28)| a) When potassium hydroxide reacts With phosphoric acid potassium phosphate and water are produced The balanced equation for this reaction is 3KOH(aq) + H;PO (aq) K;POA (aq) + 3H20() If